The ELGS organizes special programs each summer on current topics related to the fields of law and governance, which draw on key experts from throughout the world.
Through the programs, participants are granted the opportunity to explore the political, institutional, regulatory and supervisory frameworks of evolving fields of law, primarily related to European and International integration.
THREE SUMMER COURSES CURRENTLY OPEN FOR APPLICATIONS:
ELGS Summer Intensive – Global Administrative Law in practice
The War in Ukraine and the Prospect of EU Enlargement
Regional and Global Implications
OPEN FOR APPLICATIONS
Academic Supervision: Professor Sabino Cassese, former Judge, Constitutional Court of Italy with the support of Professor Edoardo Chiti, University of La Tuscia, Italy
22-26 July 2024 – ELGS campus Legrena, Sounion GR
This week-long summer program is focused on the latest global and regional reactions to the war in Ukraine. On 14 December 2023, the EU’s highest political organ, the European Council, reiterated its dedication and determination “to further weaken Russia’s ability to wage its war of aggression” and concomitantly “to continue to provide strong political, financial, economic, humanitarian, military and diplomatic support to Ukraine and its people for as long as it takes.” A similar stance has been adopted by other international actors such as NATO, the UN and the OECD. Alas, upholding this stance may provoke a series of consequences, which may heavily impact not only Russia, but also EU States and third States. Being a multi-faceted phenomenon, an inter-State conflict of this magnitude has tested the adaptability of international institutions to a new international reality.
By placing a special emphasis on the EU as a global actor, this ambitious program will zoom in five key areas of interest, with a view of assessing how effective international institutions have been so far in addressing the repercussions of the Ukraine war and whether their reform is in order to administer relevant future challenges. In particular, the five areas of interest are: Europe’s energy crisis, EU economic and monetary policy, sanctions, EU enlargement, and finally humanitarian assistance.
The GAL in action summer program aims to attract a disparate cohort of participants; mature students in law (LLM, PhD) and political science (MA, PhD) that have an interest in international institutions and their functioning, early-stage practitioners engaged in the respective fields, policy-makers and domestic administrations who seek ways to enhance their professional skills by keeping up-to-date with global regulation issues.
Designed for:
This is a generic course addressed to undergraduate and post-graduate students in law, economics and political science, as well as civil servants and legal practitioners: legal advisors, lawyers and officials of international organizations and domestic administrations who seek ways to enhance their professional skills by keeping up-to-date with emergent global regulation issues.
The participants will attend courses in the various fields conducted by eminent professors from prestigious Universities from all over the world. All the courses will be seminar-like classes and colloquia, and the active class-participation will be encouraged.
Cost of the Program:
The cost is 490€.
Applications deadline: June 14, 2024
How to apply: Applicants should submit an updated CV and letter of motivation in English to summerschools@elgs.eu Information at T: +30 211 311 0 671
Program 2024 This year the program will run on premise from 22 to 26 July from 10.00-17.00, under the title “Global Administrative Law in Practice”. Ιt will include one day of study visit in Athens and other social and cultural events.
2024 PROGRAM and INSTRUCTORS soon to be announced.
FACULTY for the 2023 Program
Professor Maurizia de Bellis
University of Rome Tor Vergata
Professor George Pagoulatos
Director General of the Hellenic Foundation for European and Foreign Policy (ELIAMEP), Athens University of Economics and Business (AUEB), Visiting Professor at the College of Europe in Bruges.
Dr. Diego Valiante
Senior Officer/Team Leader at European Commission & Adjunct Professor at Università di Bologna
Dr. Konstantina Georgaki
Policy Officer at European Commission (DG FISMA), Brussels
Fellow, European Law & Governance School, Greece
You can download the BROCHURE here.
Dr. Giovanni Zaccaroni, Brexit Institute of the Dublin City University, Ireland during the online lesson
Click here to see Previous Sessions
2023 Program
Reforms of Economic Governance In the European and Global Context
Academic Supervision: Professor Sabino Cassese, former Judge, Constitutional Court of Italy with the support of Professor Edoardo Chiti, University of La Tuscia, Italy
24-28 July 2023 – ELGS campus Legrena, Sounion GR
With the publication in November 2022 of a Communication setting forth its orientations for a review of the EU economic governance framework, the European Commission has resumed a long-going debate among policymakers and academics and triggered a remarkable process of reform, destined to shape the EU economic governance for the next decade.
This week-long summer school provides a unique forum for a critical discussion on such process of reform. The course will open by situating the ongoing reform of the EU economic governance framework in its wider economic and political context. It will then address the key institutional and legal issues of the new framework for EU’s economic policy coordination and surveillance. Specific attention will be paid to the new policy modes, implementing and enforcement mechanisms, and accountability tools, all considered as crucial institutional innovations of the EU and global legal landscape. The idea is to discuss the reform of the EU economic governance framework both from a theoretical and practical perspective, by making use of active learning methodologies and relying on participants’ involvement in the discussion of the main legal and institutional issues at stake.
2023 PROGRAM
The program ran on premise from 24 to 28 July from 10.00-17.00, under the title “Global Administrative Law in Practice: Reforms of Economic Governance, in the European and Global Context”. Ιt included one day of study visit in Athens and other social and cultural events.
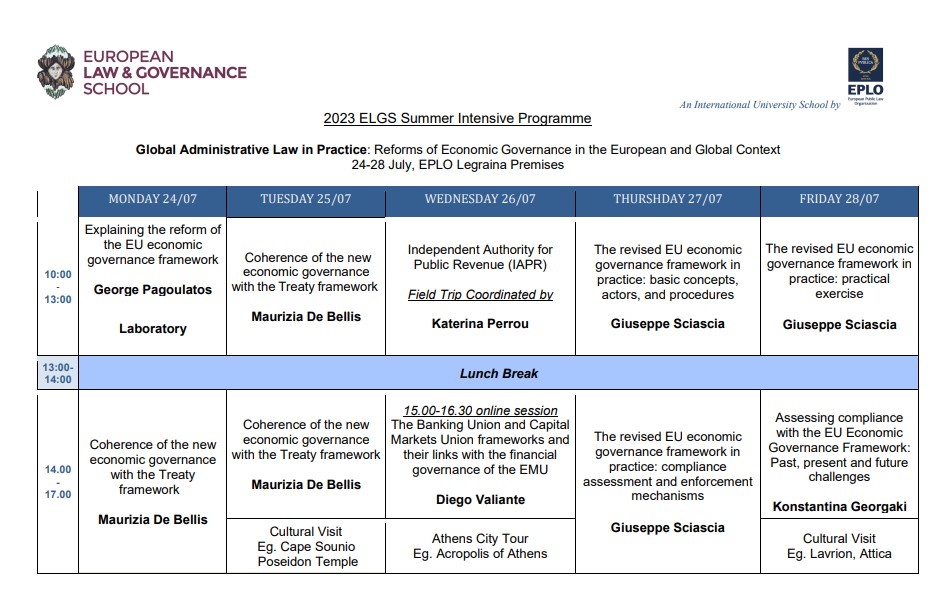
- INTRODUCTION 24 March, 2023 “Explaining the reform of the EU economic governance framework” Professor George Pagoulatos, Director General of the Hellenic Foundation for European and Foreign Policy (ELIAMEP), Professor of European Politics and Economy at the Athens University of Economics and Business (AUEB), and Visiting Professor at the College of Europe in Bruges.
- PART 1 “Coherence of the new economic governance with the Treaty framework” Prof. Maurizia De Bellis, Università di Roma Tor Vergata
- PART II “The revised EU economic governance framework in practice: basic concepts, actors, and procedures. Compliance assessment and enforcement mechanisms. Practical exercise” Dr. Giuseppe Sciascia, Policy Officer at the European Commission – DG REFORM
- Online guest lecture by Dr. Diego Valiante, Senior Officer/Team Leader at European Commission & Adjunct Professor at Università di Bologna.Topic: “The Banking Union and Capital Markets Union frameworks and their links with the financial governance of the EMU”.
- Guest Lecture in situ by Dr. Konstantina Georgaki, Policy Officer at European Commission (DG FISMA), Brussels, Resident Lecturer, European Law & Governance School, Greece. Topic: “Assessing compliance with the EU Economic Governance Framework: Past, present and future challenges”.
- Field trip, coordinated by Dr. Katerina Perrou, Post-Doctoral Fellow, National and Kapodistrian University of Athens Law School, Legal Counsel to the Governor of the IAPR at the Independent Authority for Public Revenue (IAPR)
2022 PROGRAM
- Is there a Global Constitution Prof. Avv. Elisa. D’Alterio University of Catania, Italy
- Global Financial Regulation Prof. Maurizia De Bellis, Tor Vergata, University of Rome
- Governing by the numbers Prof. Marta Infantino, University of Trieste, Italy
- The administrative State under Siege Prof. Elisabetta Morlino, University Suor Orsola Benincasa of Naples, Italy
- Brexit Dr. Giovvani Zaccaroni, Lecturer, Department of European and Comparative Law, University of Malta
GUEST INTERVENTIONS
Human Rights in Europe, Linos-Alexander Sicilianos, Professor, Dr, Judge, Dean of Athens Law School, National and Kapodistrian University of Athens, former President of the European Court of Human Rights and Guido Raimondi, Professor, former President of the European Court for Human Rights;
Panel moderator: Professor Avv. Elisa. D’Alterio
Global regulation in banking and Trade law, Christos Gortsos, Professor, Law School, National Kapodistrian University of Athens
Giorgio Sacerdoti, Emeritus Professor Bocconi University, Italy, former chairman of the WTO AB
Panel moderator: Professor Maurizia De Bellis
2020 Program – Administrative Law in Practice
- Is there a Global Constitution?- Lorenzo Casini, IMT School for Advanced Studies, Lucca
- The Administrative State under Siege –Elisabetta Morlino, Universitá Suor Orsola Benincasa
- Governing by the numbers, Prof. Marta Infantino, University of Trieste, Italy
- Global financial regulation, Prof. Maurizia De Bellis Tor Vergata, University of Rome
- Brexit, Dr. Giovanni Zaccaroni, Brexit Institute of the Dublin City University
2019 Program ran under the title: Comparative Law and Global Governance
- Is there a Global Constitution?- Lorenzo Casini, IMT School for Advanced Studies, Lucca
- The Administrative State under Siege –Elisabetta Morlino, Universitá Suor Orsola Benincasa.
- The Εvolution of EU Energy Market Regulation- Aikaterini Ιliadou , Professor at the National and Kapodistrian University of Athens
- Judicial Review: Old and New Problems- Diletta Tega, Bologna University
- Where is the European Union going?– the future of the EMU, Christos Gortsos, Law School of the National and Kapodistrian University of Athens
- Where are we with Brexit? – Federico Fabbrini, School of Law and Government of Dublin City University
- The Dialogue of Courts – Diletta Tega, Bologna University
- Markets and States in Global Governance – Eser Karakas, University of Strasbourg
- Allocation of International Responsibility between member States and International Organisations: tug-of-war or passing the buck?, Nikolas Voulgaris, European Law and Governance School
- The autonomy of the EU legal order vis-à-vis international law, Cristina Contartese, European Law and Governance School
of Naples
Prof. Lorenzo Casini, IMT School for Advanced Studies, Lucca











MIGRATION SUMMER SCHOOL (MIGSS) 2024
“Women in International Migration”
Addressing ethnic-racial discrimination, socioeconomic and health inequalities and gender-based violence
22-27 July, 2024
Athens and Sounion – In situ
In collaboration with MEDMA – An agency of the EPLO
- Α 6-day program in English will investigate the developments in challenges regarding ethnic and racial inequalities, health disparities and precarity, integration and social exclusion of migrant and refugee women in reception societies.
- Open for Practitioners, NGO Professionals, Researchers & Post-Graduate Students of all disciplines (i.e. Sociology, Social Policy, International Relations, Human Rights, and Economics et al.)
- Meet key academics and experts working in the field
- On-site visits to Accommodation facilities and NGOs
- Doctoral students and researchers will have the opportunity to present their PhD/research at the PhD Seminar
The Context
- The presence of women in migration flows is not a new element, as females have always been an important component of international migration. Women are migrating alone, as heads of households and also as economically active individuals, while fewer than in the past are migrating as dependents of their spouses. This development of international migration has been recorded in the literature under the term “feminisation of migration” (Castles & Miller, 1998:16). Forms of female migration include those who migrate alone or before their husbands, as both the labour market and the gender division of labour in reception countries offer them better employment and economic prospects, notwithstanding severe limitations in their rights. In this context, the course 8th Migration Summer School (MIGSS): “Women in International Migration: Addressing ethnic-racial Discrimination, socioeconomic and health inequalities, and gender-based violence” has been carefully designed and developed to provide participants with a comprehensive understanding of the impact of investigating the impact of women’s migration trajectory, social networks and contextual conditions on their integration or marginalisation in host societies. This will be achieved through lectures and discussions led by international scholars, professors, and experts and study visits. This program is organized by the European Public Law Organization (EPLO). The MIGSS will take place from 22 July – 27 July 2024.
About the course
This 6-day course 8th Migration Summer School (MIGSS): “Women in International Migration: Addressing ethnic-racial discrimination, socioeconomic and health inequalities and gender-based violence” will investigate the impact of women’s migration trajectory, social networks, and contextual conditions on their integration or marginalisation in host societies.
Addressed to
As this program is interdisciplinary by nature, it is open to all disciplines (such as Sociology, Social Policy, International Relations, Human Rights, Economics et al.) and applicants from an undergraduate level of studies and beyond are welcome, including but not limited to postgraduate students, PhD candidates, researchers, and NGO professionals. Doctoral students and researchers will have the opportunity to present their PhD/research at the PhD Seminar, provided a minimum of five students are enrolled in the seminar.
Certificate
Participants will be awarded a certificate of attendance at the end of the program, after demonstrating their full and active participation throughout the 6-day course. The program does not require written work to be submitted or exams to be taken.
Credits info
8 ECTS: Participants who wish to obtain ECTS are required to submit an essay of approx. 5.000 words at the end of the Summer School on a topic related to the lectures of the MIGSS and supervised by a lecturer of their choice. Deadline for essay submission: 1st September 2024.
Languages
The Summer School program is offered in English.
Cost
550€ which includes: tuition fees, transportation costs from the hotel in Sounion to the lecture hall, study visits arrangement, and transportation.
How to Apply
Applicants should submit an updated CV and letter of motivation in English to summerschools@elgs.eu, T: +30 211 311 0 671 by June 28, 2024.
Practical Information:
Accommodation
Students are invited to secure their accommodation and transportation or may opt for an accommodation package that is provided through the EPLO. Participants will be accommodated at the seaside premises of the EPLO at Sounion, Greece, or at a partner hotel. Accommodation is on a first-come-first-serve basis and students interested in this package should inquire at summerschools@elgs.eu
Facilities
The program takes place at EPLO facilities in Sounion, one hour away from Athens by car, overlooks the Aegean Sea, and is located by the bay of Cape Sounion, where the ancient Temple of Poseidon rests. Participants will have access to EPLO facilities, which include a resource library, study room, and sports facilities.
Students are encouraged to bring a laptop in order to study relevant references and reading material including statistics, maps, reports, scientific articles on aspects of the phenomenon of migration and refugee flows, and presentations used by the speakers.
2024 Instructors
Soon to be announced.
2023 Instructors:

Head of the Drug Department of the General State Chemistry (ret.) – Vice president P.A.D.A.
Assistant Professor, Sociology and Social Policy, Centre for Interdisciplinary Research into Health and Illness, University of Wroclaw Poland.
Assistant Professor, Department of Public Health Policy, School of Public Health, University of West Attica, Greece
Anthropologist, Researcher, Associate Member,
Centre d’études en sciences sociales sur les mondes africains, américains
et asiatiques, Université de Paris, France.

Professor, Department of Sociology, Panteion University of Social and Political Sciences, Director of METHEXI Lab

Supervising Psychiatrist, Mental Health Center of Sotiria General Hospital – P.A.D.A. Member

Sociologist, Researcher at GEDIME/CER-Migracions, Associate lecturer, Universitat Autònoma de Barcelona, Spain
Assistant research fellow, University of Szeged, Institute of Public Law,
Consultant, Constitutional Court, Hungary
Director of Social Integration, Ministry of Migration and Asylum, Greece.
2024 Migration Summer School Schedule

Session 2023
MIGRATION SUMMER SCHOOL (MIGSS) 2023
“Migration and Asylum in Times of Crises”
24-30 July, 2023
Will run ON PREMISE
This year with the support of Mathias Corvinus Collegium (MCC), Hungary.
- Α week-long program in English will investigate the developments, challenges, integration and social exclusion of migrants and refugees in Europe as well as in reception societies.
- Open for Practitioners, NGO Professionals, Researchers & Post-Graduate Students of all disciplines (i.e. Sociology, Social Policy, International Relations, Human Rights, and Economics et al.)
- Meet key academics and experts working in the field
- On-site visits to Reception facilities and NGOs are part of this program.
The structure of the Program
- The summer school gives participants a theoretical and practical understanding of the current situation of migrants, asylum seekers and refugees during the COVID-19 pandemic, with particular focus on the current situation in Europe. This will be achieved through lectures and discussions led by internationally acknowledged professors and experts. This program is organized by the European Public Law Organization (EPLO), and will operate from the EPLO premises at Legrena Sounion. MIGSS is scheduled to run this summer IN SITU with CLASSROOM courses and study visits.
Context
- Both migration and asylum issues are central topics in national and global governance. The rise in the number of international migrants has been unrelenting over the last twenty years, reaching 281 million people living outside their country of origin in 2022 (United Nations, 2022). An extensive range of reasons and forces have led to unprecedented levels of migration, ranging from safety seeking, persecution, military, and civil conflicts, as well as political and economic changes. Since 2015, several global migration and refugee crises have emerged, including immense flows of asylum seekers and migrants to Europe’s shores and major displacements from new and ongoing conflicts. Many shipwrecks and tragic deaths prompted for an urgent policy response on behalf of the European Union with particular focus on search-and-rescue operations and thwarting the migrant smuggling industry and trafficking networks. Child migrants and unaccompanied minors traveling alone towards the EU or the United States encountered multiple dangers including the risk of abuse and trafficking. Along with the escalation of the flows the EU was confronted with five interrelated crises: a) A Central, Eastern Mediterranean migration, border-control and humanitarian crisis; b) A growing asylum and protection crisis as national asylum mechanisms and legal systems had to deal with mixed flows of many nationalities, including those to be granted protection, e.g., Syrians, Iraqis, Afghans, and others; c) Congested accommodation facilities, stressful and unhygienic conditions; d) Public opinion of local societies curved from positive views to greater skepticism resulting in the growing power of the political extreme right along with an anti-immigrant rhetoric leading towards a crisis of solidarity; e) Overstressed integration processes. Natural disasters highlighted the connection between a changing climate and human mobility, leading to extended displacements in 2020 global displacement increased dramatically with 82.4 million (UNHCR, 2020) individuals worldwide displaced internally or externally as a result of persecution, conflict, violence, human rights violations in the Central African Republic, Afghanistan, South Sudan, Ethiopia, Burkina Faso, Syria, Myanmar, Ukraine, Somalia, Venezuela and Yemen, suffering dire conditions and clashes (European Commission, 2021). During 2020 and 2021 amid the Covid-19 pandemic, wider disputes regarding international burden-sharing and humanitarian protection emerged. Beyond restrictions on international mobility and legal migration, changes to informal migration routes were sparked (MPI, 2020). Immigrants have been among the worst hit by the pandemic and migrant workers were at a higher risk of being infected since they were in precarious employment and under a precarious legal status, while they were considered essential workers throughout the public-health crisis in both low- and high-skilled jobs. Reception countries differ immensely regarding their capacity, perceptions, and practices to integrate migrants and refugees financially and socially into their societies. Thus, a global framework for handling migration crises is required.In this context, the course 7th Migration Summer School (MIGSS): “Migration and Asylum in Times of Crises” has been carefully designed and developed to provide participants with a comprehensive understanding of the impact of multiple crises on the reception, management, human rights and integration of immigrants, asylum seekers and refugees. The summer school gives participants a theoretical and practical understanding of the current situation of migrants, asylum seekers and refugees. This will be achieved through lectures and discussions led by internationally acknowledged professors and experts and study visits. This program is organized by the European Public Law Organization (EPLO) from 24-30 July 2023.
Certificate
Participants will be awarded a certificate of attendance at the end of the program, after demonstrating their full and active participation throughout the 7-day course. The program does not require written work to be submitted or exams to be taken.
Credits info
8 ECTS
Participants who wish to obtain ECTS are required to submit an essay of approx. 5.000 words at the end of the Summer School on a topic related to the lectures of the MIGSS and supervised by a lecturer of their choice. Deadline for essay submission: 1st September 2023.
Languages
The Summer School program is offered in English.
It is addressed to:
As this program is interdisciplinary by nature, it is open to all disciplines (such as Sociology, Social Policy, International Relations, Human Rights, Economics et al.) and applicants from an undergraduate level of studies and beyond are welcome, including but not limited to postgraduate students, PhD candidates, researchers, healthcare officials and NGO professionals.
Cost
550€ which includes: tuition fees, transportation cost from the hotel in Sounion to the lecture hall, study visits arrangement and transportation.
How to Apply
Applicants should submit an updated CV and letter of motivation in English to summerschools@elgs.eu, T: +30 211 311 0 671 by July 14, 2023 (extended).
Practical Information:
Accomodation
Students are invited to secure their own accommodation and transportation, or may opt for an accommodation package that is provided through the EPLO.
Participants will be accommodated at the seaside premises of the EPLO at Sounion, Greece or at a partner hotel. Accommodation is on a first-come-first-serve basis and students interested in this package should inquire at summerschools@elgs.eu
Facilities
The program takes place at EPLO facilities in Sounion, one hour away from Athens by car, overlook the Aegean Sea and are located by the bay of Cape Sounion, where the ancient Temple of Poseidon rests. Participants will have access to EPLO facilities, which include a resource library, study room, and sports facilities.
Students are encouraged to bring a laptop in order to study relevant references and reading material including statistics, maps, reports, scientific articles on aspects of the phenomenon of migration and refugee flows and presentations used by the speakers.
Session 2022
Migration Summer Schools 2022 “Rethinking the governance of migrant and refugee integration at local level”
Hybrid Format, 25-31 July, 2022
- The summer school gives participants a theoretical and practical understanding of the current situation of migrants, asylum seekers and refugees during the COVID-19 pandemic, with particular focus on the current situation in Europe. This will be achieved through lectures and discussions led by internationally acknowledged professors and experts. This program is organized by the European Public Law Organization (EPLO), and will operate online by EPLO. MIGSS is scheduled to run this summer both IN SITU with CLASSROOM courses and ONLINE with synchronous e-learning and with relevant adjustments to the program and a slightly reduced cost. Those who wish to participate in the online mode of the program they should follow the application process as is for the taught program but make sure to state clearly that they apply for the online program.
Context
- The rise in the number of international migrants has been strong over the last twenty years, reaching 281 million people living outside their country of origin in 2020, an increase from 173 million in 2000 and 221 million in 2010, while currently, international migrants represent about 3.6 per cent of the world’s population. An extensive range of reasons and forces have led to unprecedented levels of migration, ranging from seeking safety, persecution, arm and civil conflicts, political and economic changes. Thus, a global framework for handling migration crises is required as the international response to the ongoing migration and refugee reception crisis was late, unsatisfactory and excessively cautious by many countries. Humanitarian tragedies along with globally publicized images of suffering and death led to the acceleration of the international co‑ordination. Since 2015 a series of global migration crises emerged, including immense flows of asylum seekers and migrants to Europe shores and major displacements from new and ongoing conflicts. Asylum seekers and migrants arrived irregularly on the shores of the frontline states Italy, Greece and Spain overwhelmed, while multiple lives lost in their efforts to cross the Mediterranean consisting a massive humanitarian crisis. A large number of shipwrecks and tragic deaths prompted for an urgent policy response on behalf of European Union and its Member-States with particular focus on search-and-rescue operations and combat of smuggling industry of migrants and trafficking networks. At the same time, child migrants/unaccompanied minors traveling alone towards the EU or the United States encountered multiple dangers including the risk of abuse and trafficking. Integration describes an individual or group process that seeks to adapt to a new country and the reality of immigrants, applicants and beneficiaries of international protection. It is a dynamic, two-way process of mutual accommodation by all immigrants and residents of EU Member-States and the promotion of fundamental rights, non-discrimination and equal opportunities for all are key integration issues. Social integration is defined as the process of interaction and mutual adaptation migrants, applicants and beneficiaries of international protection as well and the host society with the aim of creating societies with strong cohesion and achieving coexistence in terms of peace and mutual understanding, while particular emphasis should be given at local level integration. The integration of immigrants, applicants and beneficiaries of international protection is a special area and an integral part of a state’s migration policy and the wider social policy for its entire population. It depends directly on the migration management policy and requires targeted and positive actions in order for the specific population to be re-socialized in the country of his/her settlement. In this context, the course 6th Migration Summer School (MIGSS): “Rethinking the governance of migrant and refugee integration at local level” has been carefully designed and developed to provide participants with a comprehensive understanding of the impact of multiple crises on the integration of immigrants, asylum seekers and refugees. The summer school gives participants a theoretical and practical understanding of the current situation of migrants, asylum seekers and refugees. This will be achieved through lectures and discussions led by internationally acknowledged professors and experts. This program is organized by the European Public Law Organization (EPLO), and will operate online by EPLO. This School will take place from 25thJuly to 31st July, 2022.
2022 Instructors
Giovanna Campani, Professor, Department of Education and Psychology, University of Florence (UniFI), Italy.
Teresa Terrón-Caro, Professor, Department of Education and Social Psychology, Vice Dean of Quality and Teaching Coordination, Faculty of Social Sciences, University Pablo de Olavide, Sevilla, Spain.
Theodoros Fouskas, Assistant Professor, Department of Public Health Policy, School of Public Health, University of West Attica, Greece.
Gerassimos Karabelias, Professor, Department of Sociology, Panteion University of Social and Political Sciences, Director of METHEXI Lab.
George Tsobanoglou, Professor, Department of Sociology, University of the Aegean, Director of Sociology of Work Lab (ergon-axia), Greece
Apostolos Veizis, MD, Executive Director, INTERSOS Hellas
Anastasia Chalkia, Dr., Asylum Expert – NKUA
Zoran Lapov, Dr. in General and Social Pedagogy, Department of Education, Languages, Intercultures, Literatures and Psychology (FORLILPSI), University of Florence, Italy.
Lefteris Papagiannakis, Director, Greek Council for Refugees
Fabiola Ortega de Mora /Dr., Faculty of Social Sciences, Pablo de Olavide University, Seville, Spain
Dimitrios Georgiadis, Camp Manager of Eleonas Refugee Camp, PhD in Education and Psychology, University of Bari
Michael Gusmano, Professor, the College of Health, Lehigh University
Maurizio Ambrosini, Professor, Department of Social and Political Sciences, University of Milan
Dimitrios Georgiadis, Camp Manager of Eleonas Refugee Camp, PhD in Education and Psychology, University of Bari
Michael Gusmano, Professor, the College of Health, Lehigh University
Course Leader: Theodoros Fouskas, Assistant Professor (elected), Department of Public Health Policy, School of Public Health, University of West Attic, Greece



Session 2019
2019 MIGRATION SUMMER SCHOOL “Moving Beyond Crisis? From exclusion to integration of migrants and refugees in Europe”
22-27 July 2019
The Structure of the Program
In this context, the course 3rd Migration Summer School (MIGSS) titled “Moving Beyond Crisis? From exclusion to integration of migrants and refugees in Europe” has been carefully designed and developed to provide participants with a comprehensive understanding of issues regarding the integration of immigrants and refugees in Europe with emphasis on the current situation in Europe.
The summer school gives participants a theoretical and practical understanding of contemporary migration in Europe, with particular focus on the current situation in Europe. This will be achieved through lectures and discussions led by internationally acknowledged professors and experts, as well as through a day of site-visits to facilities and organizations in Athens that support refugees, and immigrants. This program is organized by the European Public Law Organization (EPLO), and will operate out of the EPLO’s premises at Sounion, Greece. This School took place from 22nd July to 27th July, 2019.
Goals
This program will offer participants insights into migration in Europe, highlighting its growing relevance for the worldwide discussion on migration and refugee integration policies and combating social exclusion in Europe. It will help them develop a broad theoretical and practical understanding of contemporary migration in Europe.
On a scientific level the program aims to develop arguments in relevance to social exclusion and integration in the context of immigrant and refugee movements, to assess the causes and effects of the migration and refugee crisis on European societies of reception and on the migrant populations themselves and to provide motivation for further research in the field.
2019 Program
Bridget Anderson, Professor of Migration, Mobilities and Citizenship, University of Bristol, United Kingdom.
Nadina Christopoulou, Ph.D. University of Cambridge, Anthropologist, and Co-founder/Coordinator of Melissa Network
Daniel Esdras, Special Envoy of the DG of the IOM, Advisor to the Regional Director for the Mediterranean.
Theodoros Fouskas, Sociologist, Ph.D., University of West Attica (UNIWA) and EPLO, Greece.
Lefteris Papagiannakis, Dep. Mayor for Immigrants, Refugees and Municipal Decentralization Municipality of Athens, Greece.
Paraskevi Gikopoulou, PhD, University of Warwick, Sociologist, External Research Associate, Department of Public Health Policy, University of West Attica, Greece
Tina Stavrinaki, Lawyer, Ph.D. University Paris 2, Assistant Coordinator of the Racist Violence Recording Network.
Course Leader: Dr. Theodoros Fouskas
Field Trips
In conjunction with the seminars, the program includes two days of study visits in Athens and Thiva. Athens provides an ultimate setting for carrying out this program, as Athens comprises the main city of temporary or permanent stay for immigrants and refugees in the country and offers a unique environment that facilitates participants’ deeper engagement.
Participants will be given the unique opportunity to interact with immigrants, refugees and representatives of organizations that provide support to them by conducting a study visit to key locations such as :
Day 1
- Accommodation Center for Migrants and Refugees in Thiva, Central Greece
- Melissa Network: Network of Migrant Women in Greece, Promoting Empowerment, Communication and Active Citizenship, Athens, Attica
Day 2
- Generation 2.0 for Rights Equality and Diversity (G2RED) – Accommodation Facility for Unaccompanied Refugee Minors, Athens,
- Open Reception Facility of Third-Country Nationals, Eleonas, Athens
- Open Centre for migrants registered for assisted voluntary return and reintegration (OCAVRR), Athens




ELGS & IHU SUMMER ACADEMY ON ARTIFICIAL INTELLIGENCE
Thematic 2024: “ENERTECH – Mastering Advanced Technologies in Energy”
1-6 July, 2024
In collaboration with the International Hellenic University
OPEN FOR APPLICATIONS.
Are you fascinated by the future of energy? Do you dream of a world powered by clean, sustainable solutions? Join us at the EnerTech Summer Academy in Athens, Greece, and unlock your potential as a changemaker in the energy revolution!
Join the Summer Academy that is taking place at the EPLO premises in Athens this year in cooperation with the Hellenic International University
- 5 days immersed in the heart of cutting-edge energy tech: Explore the latest advancements in renewable energy, smart grids, microgrids, hydrogen, nuclear energy and more.
- Dive deep into diverse topics: From entrepreneurship and policy to law and new technologies, gain a comprehensive understanding of the modern energy landscape.
- Learn from the best: Engage with renowned professors, industry experts, and innovators who are shaping the future of energy.
- Network with a vivid community: Connect with passionate peers from various disciplines, building lifelong friendships and collaborations.
- Athens as your classroom: Experience the vibrant energy of this historic city, while exploring its ancient wisdom and modern sustainability initiatives.
More than just a summer program, this is an immersive experience that will:
- Ignite your passion: Spark your curiosity and inspire you to become an active agent in the energy transition.
- Empower you with knowledge: Equip you with the skills to tackle energy challenges.
- Connect you with a network: Build a community that shares your vision for a sustainable future.
Join us in Athens this summer and embark on a journey that will transform your perspective, ignite your potential, and empower you to shape the future of energy!
The program addresses:
• Students: Enhance your knowledge, gain practical skills, and discover career paths in the booming energy sector.
• Young professionals: Expand your expertise, network with industry leaders, and accelerate your career in energy innovation.
• Anyone with a passion for energy: Deepen your understanding of the energy challenges and contribute to building a brighter future.
Programme Structure & Working hours
The lectures will be live and will take place, in Athens, during the first week of July 2023 (between the 1st and the 6th of July 2024). More specifically, for the period of 1 week, 6 lectures will be offered from Monday 1 July to Saturday 6 July (10.00a.m.-12.00 p.m. & 12.10-14.00 p.m.). Along with the lectures, the program includes some further activities; the participants will be given the opportunity to participate to educational visits.
2024 Program and Instructors:
Monday 01/07/2024 // 10:00 – 14:00 // Decentralizing Power – Microgrids & Virtual Power Plants
Lecturer: Prof. Michael Caramanis, Boston University, former Chairman of Hellenic Regulatory Authority of Energy
Tuesday 02/07/2024 // 10:00 – 14:00 // New Horizons in Energy Entrepreneurship
Lecturer: Dr. Stelios Bikos, General Manager, Hellenic Energy Competence Center – HECC
Wednesday 03/07/2024 // 10:00 – 14:00 // The Digital Pulse of Energy Transformation
Lecturers: Prof. Nikos Chatziargiriou & Prof. Aris Dimeas, National Technical University of Athens
Thursday 04/07/2024 // 10:00 – 14:00 // Innovation Driving the Low-Carbon Energy Transition
Lecturer: Prof. Theodore Panagos, International Hellenic University, former Vice-Chairman of Hellenic Regulatory Authority for Energy
Lecturer: Prof. Dimitra Vagiona, Aristotle University Thessaloniki
Friday 05/07/2024 // 10:00 – 12:00 // Nuclear Energy and Technology
Lecturer: Prof. Nikolaos Petropoulos, National Technical University
// 12:00 // Field Trip to Revithoussa LNG Terminal
// 14:00 – 16:00 // Green H2 Routes: Crafting the Regulation and Transportation Infrastructure for Europe’s Hydrogen Transition
(Revithoussa Island/ DESFA LNG Terminal)
Lecturer: DESFA H2 Committee
Saturday 06/07/2024 // Field Trips Day – Acropolis Museum
Farewell lunch
Applications:
Applicants should submit an updated CV and letter of interest in English to summerschools@elgs.eu or contact us at +30 211 311 0 671 by 14 June 2024.
A Certificate of Attendance will be awarded at the end of the program to all attendees who have fulfilled all requirements.
Accommodation and Tuition Fees
The tuition fees are 250 € (non-refundable). The fees will cover:
- Participation in the Summer Academy
- Welcome Social Event on the Induction Day
- Transportation for a cultural/educational visit
- Goodbye Social Event (Dinner)
All participants are responsible for covering travel costs from their home cities to Athens and other everyday expenses. Participants will have to cover their travel and accommodation expenses. Participants can ask the EPLO/ELGS secretariat for the accommodation package offered.
Venue
• The program takes place in Athens EPLO Premises.
2024 ENERTECH Summer Academy Schedule

Click here to see the Previous Sessions
ELGS & IHU SUMMER ACADEMY ON ARTIFICIAL INTELLIGENCE
Thematic 2023: “METAVERSE – Ethical, Economic and Legal Challenges”
3-8 July, 2023
In collaboration with the International Hellenic University
OPEN FOR APPLICATIONS.
Dive into the exciting world of the Metaverse: Join the Summer Academy that is taking place in Thessaloniki in cooperation with the Hellenic International University and explore the ethical, economic, and legal challenges we are facing as a consequence of the rapidly evolving technology. Our academy is designed for students, professionals, and anyone interested in the Metaverse and its impact on society. You’ll learn from leading experts in the field, who will provide you with a comprehensive understanding of the technology, its potential, and its challenges.
Despite the growing popularity of the Metaverse, the concept is still unclear to many and we are still in the metaverse’s early days. Many are forecasting what the metaverse means for the future, despite not everybody being quite on board yet. In fact, in 2022 Facebook renamed itself “Meta” and CEO Mark Zuckerberg envisions that a billion people will one day be in the metaverse spending lots of money and it will generate revenue from selling virtual services and products.
But what is the metaverse, how does it work and which are its crucial ethical, economic and legal aspects? The metaverse brings with it a variety of controversies and concerns, real-world problems, safety and privacy concerns. Among others, all these issues relating to Metaverse are going to be analyzed in depth, while light will be spread on “Metaverse” in a holistic way.
Programme Structure & Working hours
The lectures will be live and will take place, in Thessaloniki, during the first week of July 2023 (between the 3rd and the 8th of July 2023). More specifically, for the period of 1 week, 6 lectures will be offered from Monday 3 July to Saturday 9 July (10.00a.m.-12.00 p.m. & 12.10-14.00 p.m.). Along with the lectures, the program includes some further activities; the participants will be given the opportunity to participate to educational visits.
The program includes lectures, case studies, and interactive discussions on the following topics:
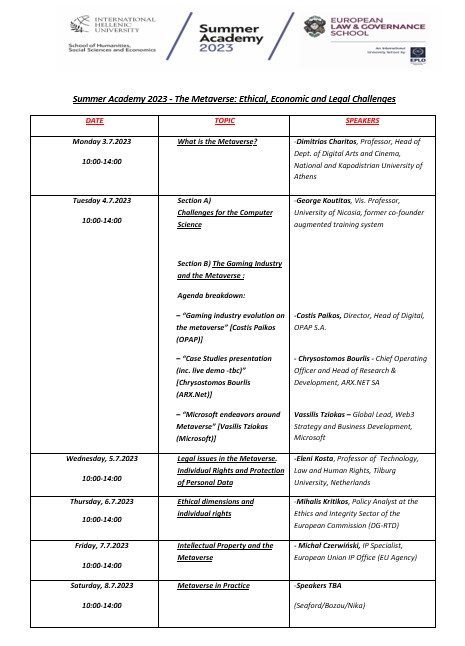
1.What is the Metaverse (3.7.2023)
• Metaverse – Challenges for the Computer Science (4.7.2023)
• Human rights challenges in the Metaverse (5.7.2023)
2. Virtual Words/Metaverse, ethics and EU policies: challenges and opportunities (6.7.2023)
3. Intellectual Property and the Metaverse (6.7.2023)
4. Metaverse in Practice (7.7.2023)
5. Cultural Visit to Thessaloniki Archaeological Museum (guided tour) (7.7.2023)
6. Farewell Lunch – Closing of Summer Academy 2023 (7.7.2023)
Download the program here
The program addresses:
The program is open to graduate students and PhD researchers from various disciplines such as Philosophy, Law, Computer Science providing them with a basic background on the metaverse, but it is also aimed at senior researchers, policy analysts, lawyers and legal experts, civil servants, members of organizations of civil society, AI practitioners and other professionals interested in expanding their understanding of AI and its impact on society.
Applications:
Applicants should submit an updated CV and letter of interest in English to summerschools@elgs.eu or contact us at +30 211 311 0 671 by 21 June 2023.
A Certificate of Attendance will be awarded at the end of the program to all attendees who have fulfilled all requirements.
Accommodation and Tuition Fees
The tuition fees are 150 € (non-refundable). The fees will cover:
- Participation in the Summer Academy
- Transportation for an educational visit/field trip.
- Goodbye Social Event (Dinner) (8th of July)
All participants are responsible for covering travel costs from their home cities to Thessaloniki and other everyday expenses. Participants will have to cover their travel and accommodation expenses. Participants can choose accommodation from a variety of options, with prices starting from 240 € for the whole week.
Venue
• The program takes place in Thessaloniki. Venue: Thessaloniki Chamber of Commerce and Industry
Thessaloniki is a vibrant city with a rich history and culture, perfect for a summer academy. You’ll have the opportunity to explore the city and enjoy the Mediterranean sea, while also gaining valuable knowledge about the Metaverse.